Energy profiles for palladium-catalysed oxidative addition of PhBr to... | Download Scientific Diagram

Developing Ligands for Palladium(II)-Catalyzed C–H Functionalization: Intimate Dialogue between Ligand and Substrate | The Journal of Organic Chemistry

Palladium(II) complexes containing different types of nitrogen ligands... | Download Scientific Diagram

Palladium-Based Metal–Ligand Assemblies: The Contrasting Behavior upon Addition of Pyridine or Acid | Journal of the American Chemical Society

Ligand-Centered Triplet Diradical Supported by a Binuclear Palladium(II) Dipyrrindione | Inorganic Chemistry

Quantifying the Donor Strength of Ligand-Stabilized Main Group Fragments | Journal of the American Chemical Society
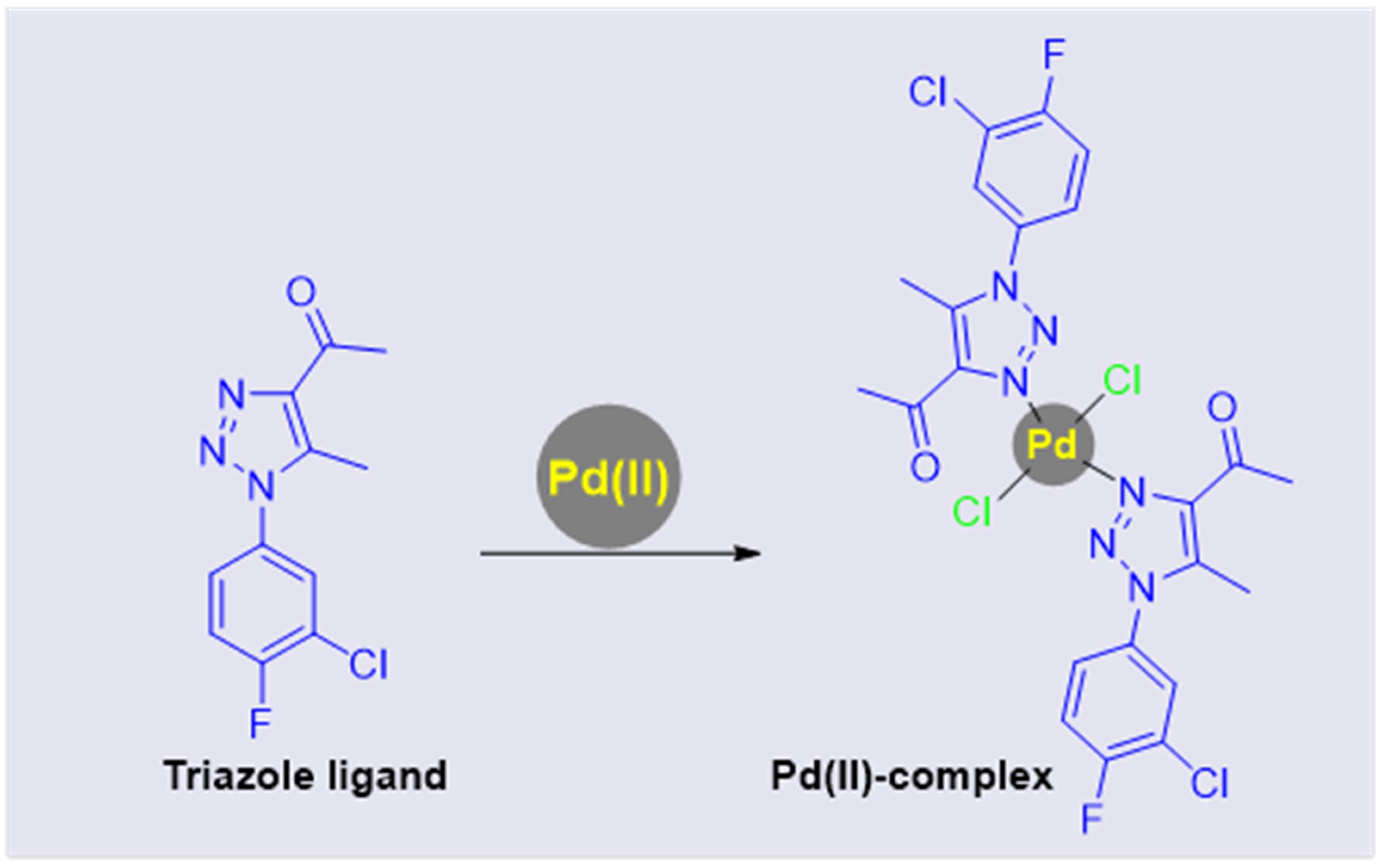
Crystals | Free Full-Text | Synthesis and Solid-State X-ray Structure of the Mononuclear Palladium(II) Complex Based on 1,2,3-Triazole Ligand

Palladium(II) complexes featuring bidentate pyridine–triazole ligands: Synthesis, structures, and catalytic activities for Suzuki–Miyaura coupling reactions - ScienceDirect
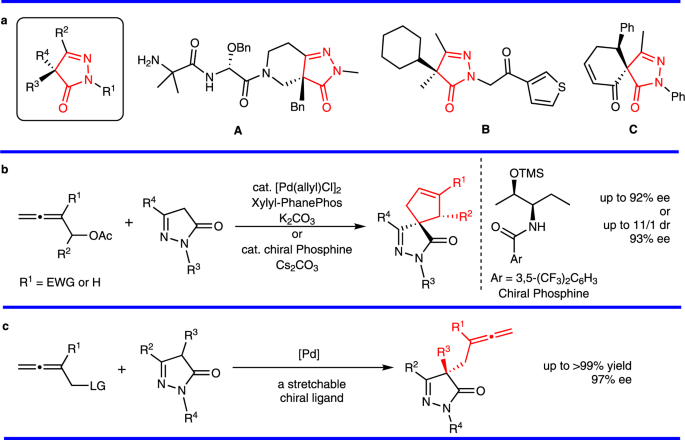
Stretchable chiral pockets for palladium-catalyzed highly chemo- and enantioselective allenylation | Nature Communications

Developing Ligands for Palladium(II)-Catalyzed C–H Functionalization: Intimate Dialogue between Ligand and Substrate | The Journal of Organic Chemistry
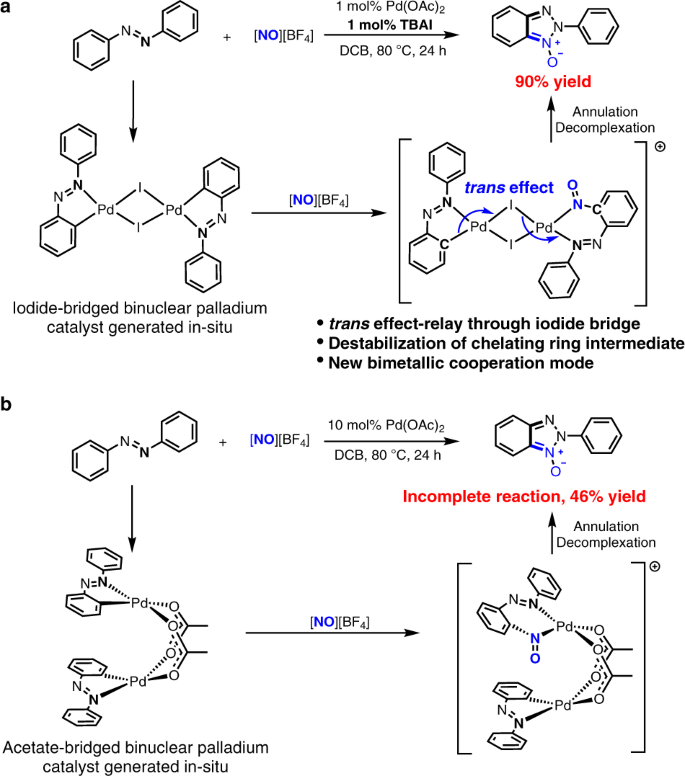
Iodide-enhanced palladium catalysis via formation of iodide-bridged binuclear palladium complex | Communications Chemistry

Palladium-catalyzed oxidative arene C–H alkenylation reactions involving olefins: Trends in Chemistry

Electrochemical Methods for Pd‐catalyzed C−H Functionalization - Erchinger - 2021 - Asian Journal of Organic Chemistry - Wiley Online Library

Phosphine Ligands in the Palladium‐Catalysed Methoxycarbonylation of Ethene: Insights into the Catalytic Cycle through an HP NMR Spectroscopic Study - de la Fuente - 2010 - Chemistry – A European Journal - Wiley Online Library

IJMS | Free Full-Text | Equilibrium Studies on Pd(II)–Amine Complexes with Bio-Relevant Ligands in Reference to Their Antitumor Activity

Developing Ligands for Palladium(II)-Catalyzed C–H Functionalization: Intimate Dialogue between Ligand and Substrate | The Journal of Organic Chemistry
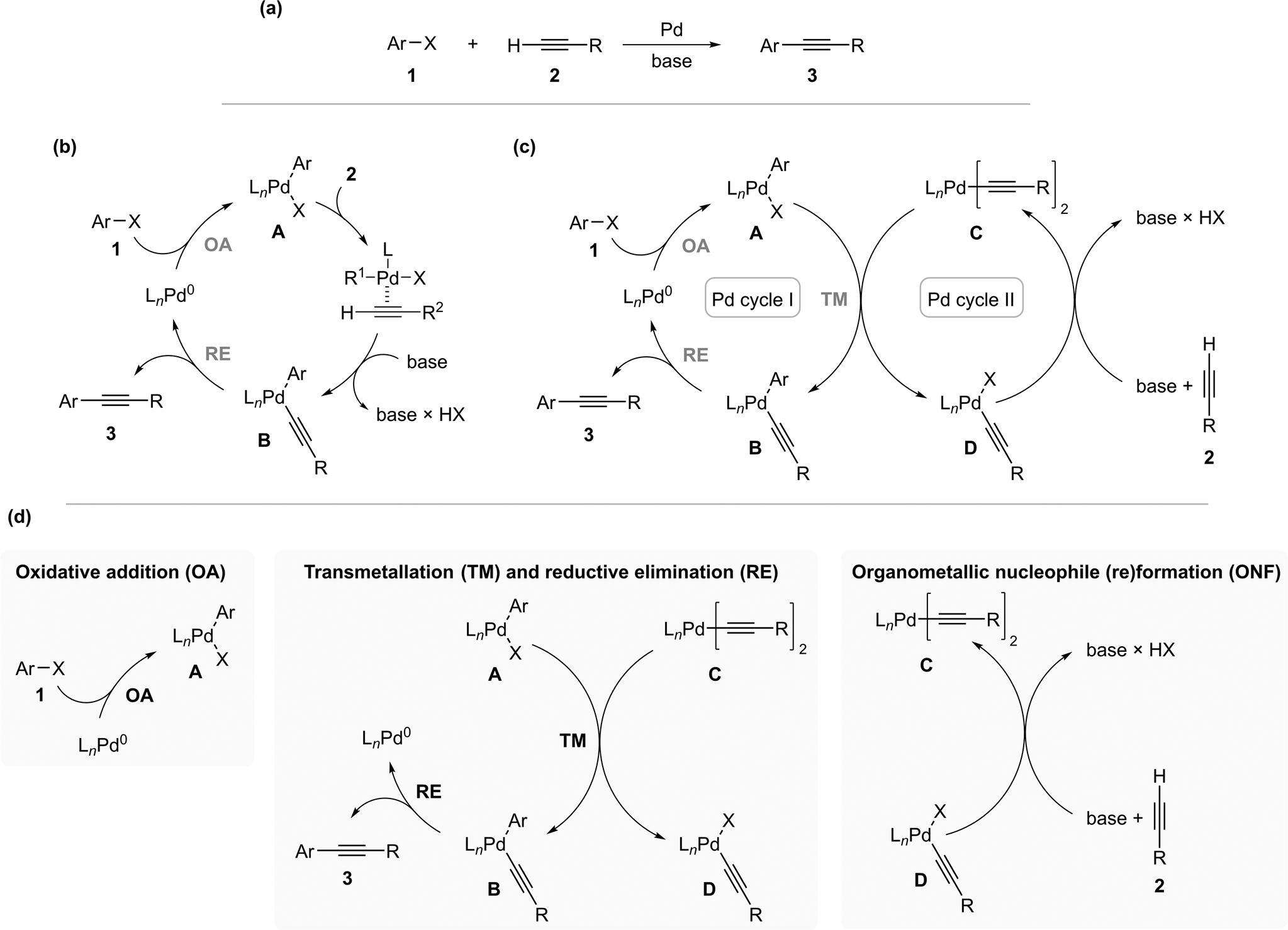
Elucidating the reaction mechanism of a palladium-palladium dual catalytic process through kinetic studies of proposed elementary steps | Communications Chemistry
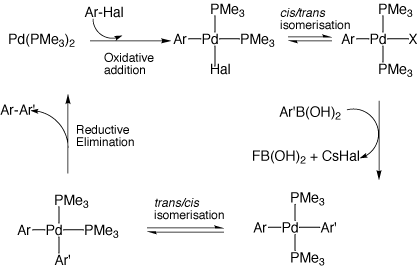
A Computational Study of the Mechanism of Palladium Insertion into Alkynyl and Aryl Carbon-Fluorine bonds

Ligand-Controlled Selectivity in the Pd-Catalyzed C–H/C–H Cross-Coupling of Indoles with Molecular Oxygen | ACS Catalysis

Biaryl monophosphine ligands in palladium-catalyzed C–N coupling: An updated User's guide - ScienceDirect

Developing backbone-modified Mor-DalPhos ligand variants for use in palladium-catalyzed C–N and C–C cross-coupling

Nitrogen ligands effects in the palladium-catalyzed carbonylation reaction of nitrobenzene to give N-methyl phenylcarbamate - ScienceDirect

Cationic Two-Coordinate Complexes of Pd(I) and Pt(I) Have Longer Metal- Ligand Bonds Than Their Neutral Counterparts - ScienceDirect

Palladium-Catalyzed C−P Bond Formation: Mechanistic Studies on the Ligand Substitution and the Reductive Elimination. An Intramolecular Catalysis by the Acetate Group in PdII Complexes | Organometallics

Donor Strength Determination of Pyridinylidene-amide Ligands using Their Palladium–NHC Complexes | Inorganic Chemistry
![Labile Pd-sulphur and Pt-sulphur bonds in organometallic palladium and platinum complexes [(COD)M(alkyl)(S-ligand)]n+—A speciation study - ScienceDirect Labile Pd-sulphur and Pt-sulphur bonds in organometallic palladium and platinum complexes [(COD)M(alkyl)(S-ligand)]n+—A speciation study - ScienceDirect](https://ars.els-cdn.com/content/image/1-s2.0-S0162013416301611-fx1.jpg)
Labile Pd-sulphur and Pt-sulphur bonds in organometallic palladium and platinum complexes [(COD)M(alkyl)(S-ligand)]n+—A speciation study - ScienceDirect

Synergistic Ligand Effect between N-Heterocyclic Carbene (NHC) and Bicyclic Phosphoramidite (Briphos) Ligands in Pd-Catalyzed Amination | Organometallics